New approach methodologies in human regulatory toxicology – Not if, but how and when! - ScienceDirect
Por um escritor misterioso
Descrição

Regulatory toxicology test guideline elements for detecting adverse

New Approach Methodologies (NAMs) for safety testing of complex food matrices: A review of status, considerations, and regulatory adoption - ScienceDirect

In Silico Toxicology - an overview

Current state of U.S. Food and Drug Administration regulation for cellular and gene therapy products: potential cures on the horizon - Cytotherapy

New approach methodologies in human regulatory toxicology – Not if, but how and when! - ScienceDirect

Toxicology in Vitro, Journal

Regulatory Toxicology and Pharmacology 44 - TRIPHASE Pharma

In silico toxicology protocols - ScienceDirect

Acceptable Intakes (AIs) for 11 Small molecule N-nitrosamines (NAs) -Pub - New Scientific Knowledge & Development - Nitrosamines Exchange
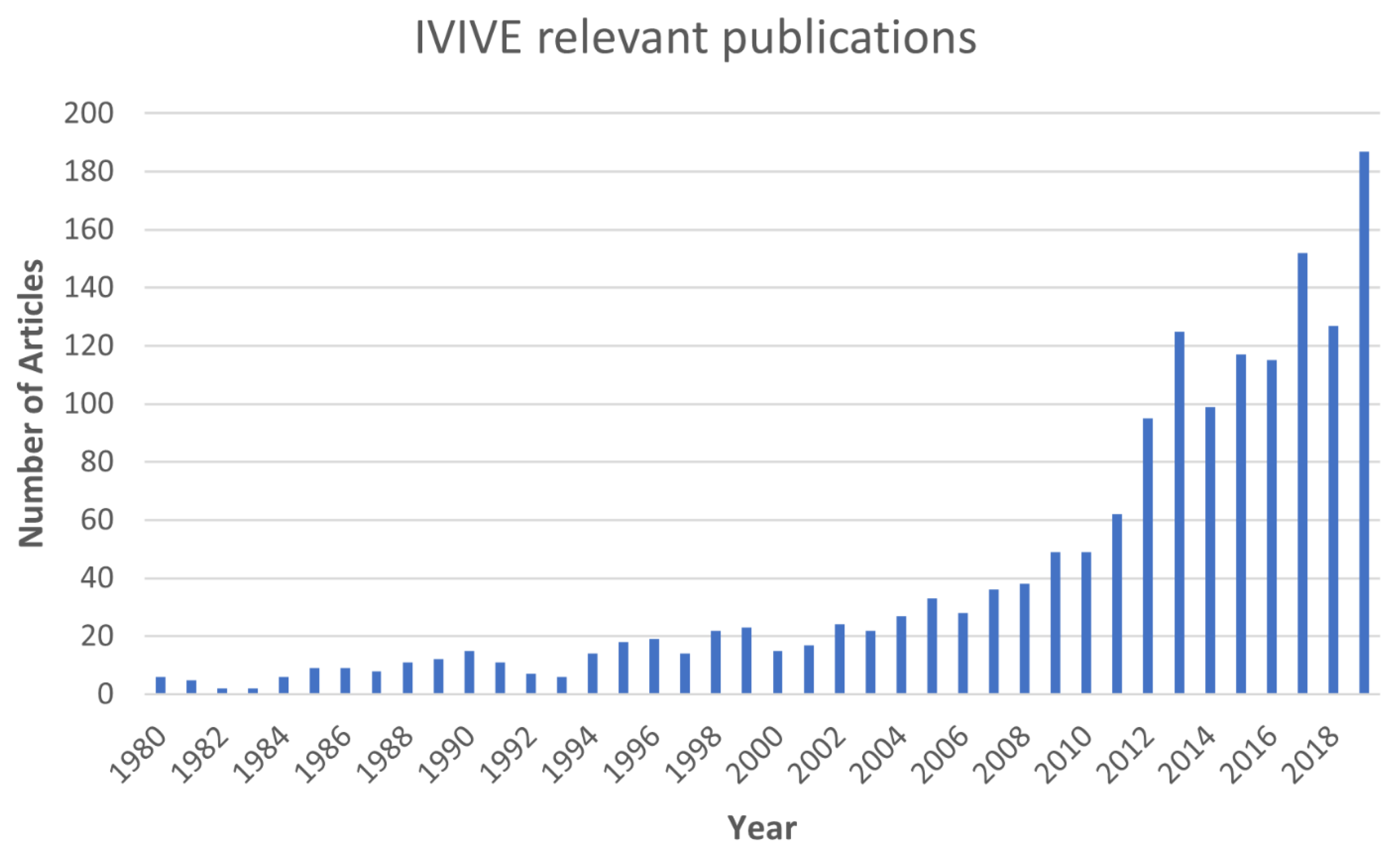
Toxics, Free Full-Text

Comparison of regulatory pathways for the approval of advanced therapies in the European Union and the United States - Cytotherapy

Suspect and non-targeted screening of chemicals of emerging concern for human biomonitoring, environmental health studies and support to risk assessment: From promises to challenges and harmonisation issues - ScienceDirect

PDF) New Approach Methodologies in human regulatory toxicology – not if, but how and when!

A Call For Action On The Development and Implementation o - 2021 - Regulatory To, PDF, Toxicology
de
por adulto (o preço varia de acordo com o tamanho do grupo)