Hep B biotech Antios closed after FDA hold proved insurmountable
Por um escritor misterioso
Descrição
Viral disease biotech Antios Therapeutics shut down earlier this year after an FDA hold on its lead hepatitis B therapy due to a serious adverse event proved insurmountable. | Viral disease biotech Antios Therapeutics shut down earlier this year after an FDA hold on its lead hepatitis B therapy due to a serious adverse event proved insurmountable.

Lab Information

Assembly Bio breaks off hepatitis B deal with Antios Therapeutics

Microfluidic Formulation of Topological Hydrogels for Microtissue

Biotech Fierce Biotech
EuroBiotech Report: Woodford's loss, BioInvent's major failure

Core Concepts - Hepatitis B Coinfection - Co-Occurring Conditions

Annalee Armstrong - Journalist Profile - Intelligent Relations
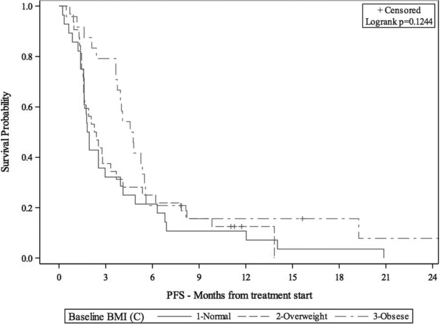
33rd Annual Meeting & Pre-Conference Programs of the Society for

Annalee Armstrong - Journalist Profile - Intelligent Relations
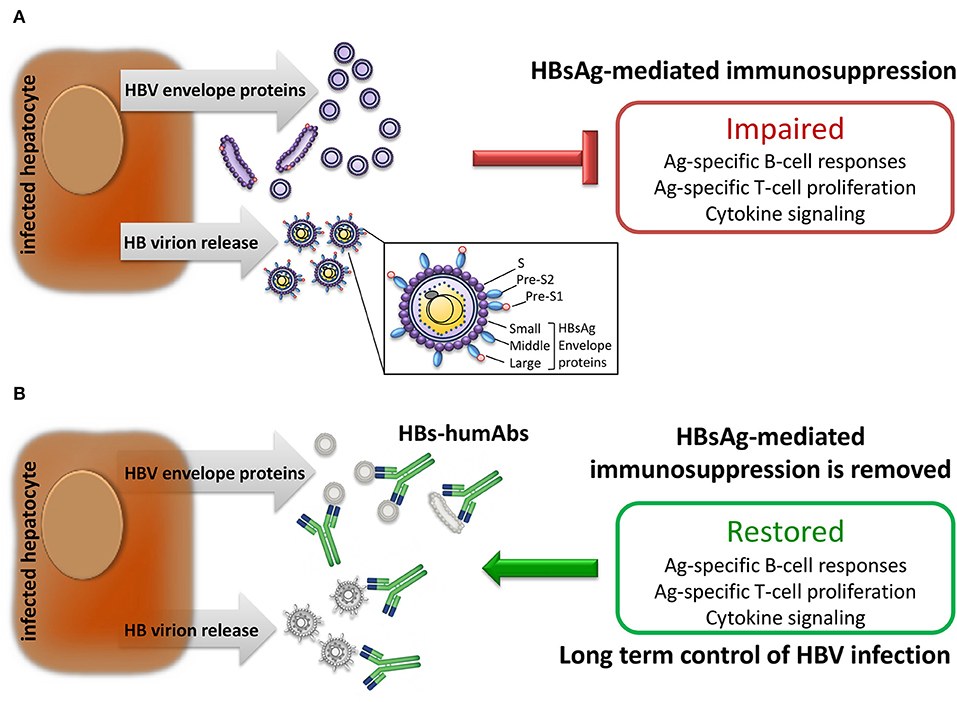
Frontiers Human Monoclonal Antibodies as Adjuvant Treatment of

LinkedIn Landon Loving 페이지: Biotech pipeline hosts 163

Immunogenicity and protective efficacy of hepatitis B vaccine

Hepatitis B Immune Globulin (Human) HyperHEP B®
de
por adulto (o preço varia de acordo com o tamanho do grupo)







